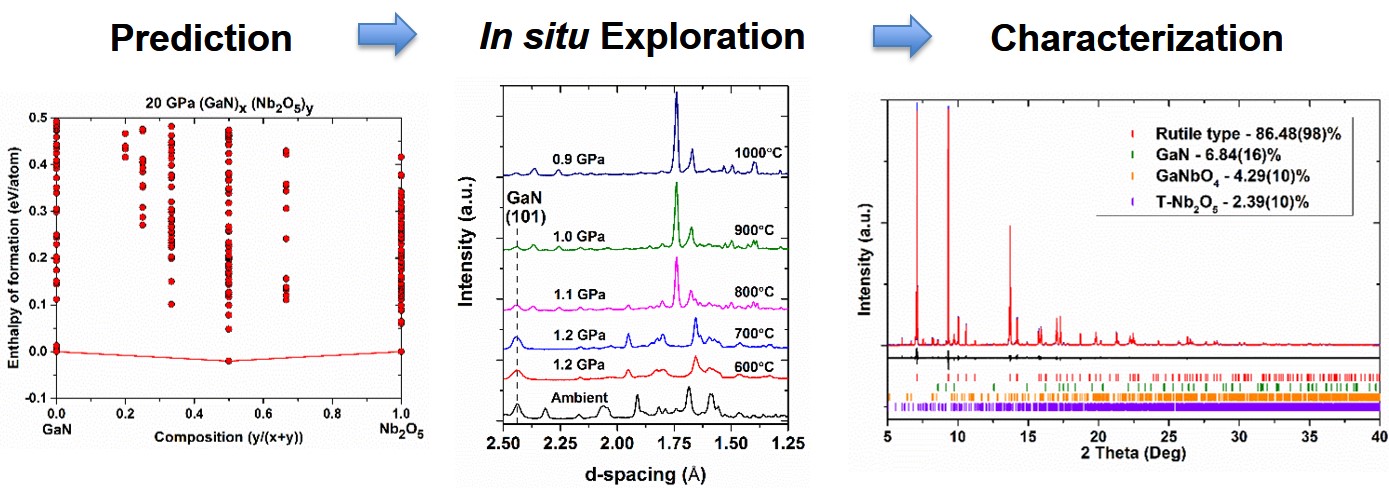
Oxynitride Semiconductors
I led a research project focused on synthesizing novel oxynitride semiconductors for photocatalytic water splitting using solid-state, high-pressure, high-temperature techniques. In order to increase the throughput of high-pressure exploratory synthesis, I developed a method of theory-guided high-pressure synthesis by combining crystal structure prediction theory using the program USPEX, in situ diffraction studies, and quench techniques. Throughout my research, I have solved and refined a multitude of oxynitride structures using a combination of X-ray and neutron powder diffraction.
Gas Adsorption in Microporous Materials
I developed an in situ X-ray diffraction – differential scanning calorimetry (XRD-DSC) technique for the rapid screening of microporous compounds for gas adsorption by simultaneously measuring enthalpy and collecting X-ray diffraction patterns to evaluate the structural response. In order to evaluate compounds for post-combustion CO2 capture, I designed and constructed a humidity atmosphere swing chamber to study the CO2 adsorption performance in simulated humid flue-gas conditions.
Mineralogy
I have conducted structural studies on crystalline, nanocrystalline, and amorphous geo-materials, primarily using X-ray diffraction with Rietveld analysis to refine structural models and perform quantitative analysis. The structural studies on crystalline materials have included synthetic akdalaite, apatites, jarosites, and phenakites, as well as solving the structure of two new minerals. Total X-ray scattering with pair distribution function analysis was used to evaluate the atomic structure of synthesized nanocrystalline ferrihydrite-related minerals and shock-induced amorphized materials.
Highlights from the structural investigations of the ferrihydrite-related mineral akdalaite. (left) The neutron powder diffraction refinement results. (middle) The complete refined structure of akdalaite with hydrogen positions and calculated bond valence numbers. (right) Pair distribution function (PDF) patterns of ferrihydrite and equivalent akdalaite.
Designing and Constructing Laboratory Equipment
XRD-DSC Gas Adsorption System
- The complete XRD-DSC system was built off a mondified Rigaku Ultima IV X-ray diffractometer. It is predominatly used for the rapid screening of novel compounds for solid-state gas adsorption. Implementation of the custom humid atmosphere swing chamber allows one to perform both vacuum and atmosphere swings up to 90% relative humidity. This was used to evaluate compounds for realistic post-combustion CO2 capture under humid conditions.
The complete XRD-DSC system.
In situ Gas Adsorption Cell
- The cell was designed and built for X-ray diffraction studies of gas adsorption in microporous compounds using either powders or single crystals. Structural investigations can be performed using any non-corrosive gas at fixed pressures between 0 and 760 torr.
The disassembled (left) and assembled (middle) gas adsorption cell with powder loaded in a quartz capillary with the manual gas loading manifold (right).
In situ Gas Adsorption Photoluminescence Cell
- The flow cell is used for preliminary testing of compounds for photoluminescence that arises from guest-framework interactions during solid-state gas adsorption. The samples are illuminated with a UV lamp as gas flows over samples in quartz tubes.
(left) The complete flow cell enclosure. (middle) Sample under UV light - the sample in air [left], under flowing carbon dioxide [middle], and an empty cell with carbon dioxide [right]. The sample under carbon dioxide emits green under short UV light. (right) The three samples one second after turning the UV lamp off revealing the sample in the flowing carbon dioxide is phosphorescencent.
Sealed Atmosphere Tube Furnace
- The tube furnace was specifically designed for high-throughput ammonolysis. The quick-clamp design of the system allows for fast sample loading and assembly. A vacuum manifold also allows for fast atmosphere evacuation and loading. The furnace sits on a dovetail slide so samples can be visibly checked during synthesis and rapidly quenched and recovered.





![(left) The complete flow cell enclosure. (middle) Sample under UV light - the sample in air [left], under flowing carbon dioxide [middle], and an empty cell with carbon dioxide [right]. The sample under carbon dioxide emits green und…](https://images.squarespace-cdn.com/content/v1/55639728e4b0f1f962341b1b/1433217372508-K9K3ZZVH6QDV98HJBPSJ/image-asset.jpeg)
